NMR Analysis of Jatropha curcas extracts.
Results shown
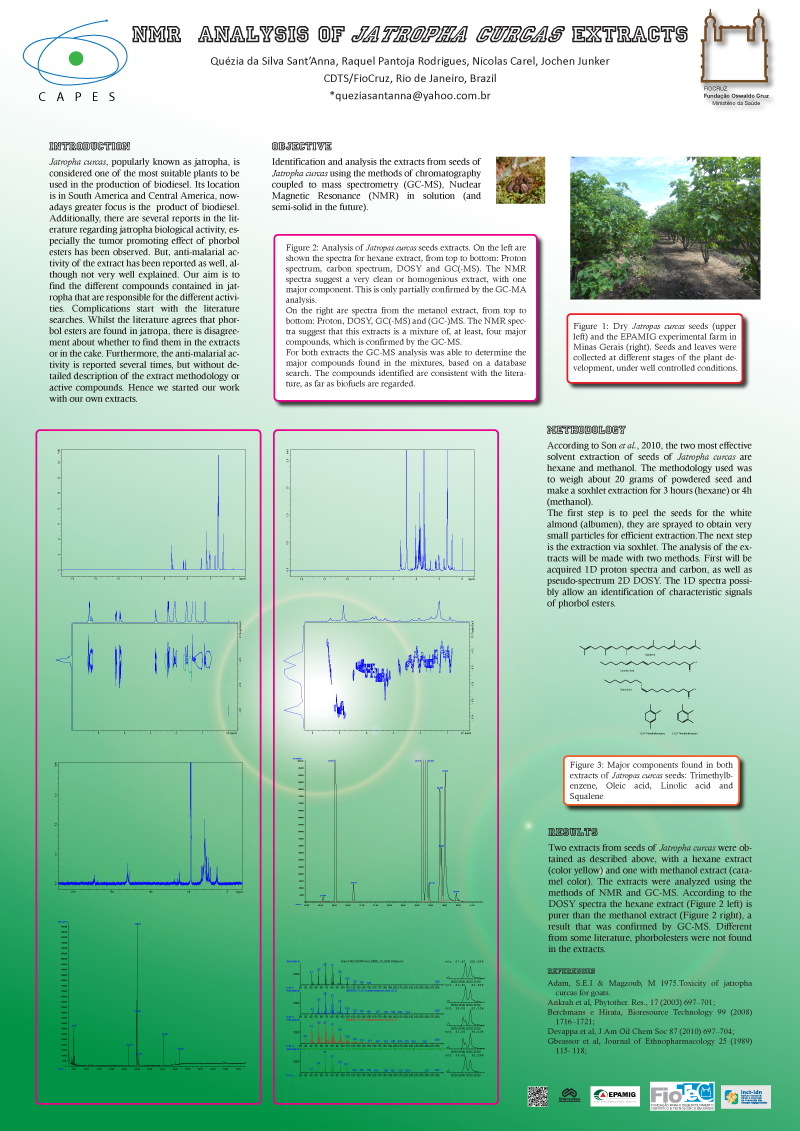
Jatropha curcas, popularly known as jatropha, is considered one of the most suitable plants to be used in the production of biodiesel. Additionally, there are several reports in the literature regarding jatropha biological activity, especially the tumor promoting effect of phorbol esters has been observed. But, anti-malarial activity of the extract has been reported as well, although not very well explained. Our aim is to find the different compounds contained in jatropha that are responsible for the different activities.
Complications start with the literature searches. Whilst the literature agrees that phorbol esters are found in jathropa, there is disagreement about whether to find them in the extracts or in the cake. Furthermore, the anti malarial activity is reported several times, but without detailed description of the extract methodology or active compounds. Hence we started our work with our own extracts.
Extracts from Jatropha curcas seeds (Son et al, 2010) were prepared using different solvents and analyzed using NMR and chromatography coupled to mass spectrometry methods (GC-MS). The 1H-NMR spectrum is very clean, showing only a small number of well-defined peaks. The 13C-NMR spectrum shows only a few peaks, as expected for fatty acids with long CH2 chains.
According to the GC-MS spectra, the major compounds found in the extracts are: Oleic and Linoleic acids (both fatty acids), 1,2,3/4-Trimethylbenzene and Squalene. None of them are reported as having biological activity. Hence, our first extracts did not reveal any compounds that could be responsible for the biological activity reported.
Literature cited
The following literature references are used for this poster:
- Adam,S.E.I.; Magzoub, M.; 1975.
- Ankrah et al; Phytother. Res. 2003, 17, 697–701.
- Berchmans; Hirata; Bioresource Technology 2008, 99, 1716–1721;
- Devappa et al; Journal of the American Oil Chemical Society 2010, 87, 697–704;
- Gbeassor et al; Journal of Ethnopharmacology 1989, 25, 115- 118;
The Poster
 English
English
 Deutsch
Deutsch
 Português
Português